News
Back row (L to R): Eric Rodrigo, Tim McNamara, Mike Lessard, Dhruv Pillai, Jon Cruz, Dallas Schray, Juri-Mikk Udam, Simon Shuham, Jack Kelly
Front row (L to R): Chuck Wehr, James Conatser, Kairn Brannon, Nick Holmes, Rosa Bonilla, Carolina Pena, Charlie Alver, Adam Jiang, Daniel Wang, Stancellous Matoreva, Brian Krentz, Vinh Nguyen
An instrument designed and built by undergraduates from the Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS) will get a lift from NASA into the upper troposphere and lower stratosphere next year to study the effects of climate change on atmospheric chemistry.
The project was among those selected for NASA’s Undergraduate Student Instrument Project grant, which awards funding to student teams to build instruments and provides the infrastructure for a guaranteed opportunity to launch them on suborbital and orbital research missions.
The team from the course “Engineering Problem Solving and Design Project” (ES 96) was awarded $200,000 for their instrument, a spectrometer that measures hydrochloric acid (HCl) in the atmosphere. The instrument will be used to better understand how complex chemical reactions erode the ozone in the stratosphere over the midsection of the United States in the summer.
ES 96 is a cornerstone of the undergraduate engineering program at SEAS. Student teams are asked to tackle a specific design problem for real-world application. Previous classes have built smokers for Williams-Sonoma, a geothermal heating and cooling system for Radcliffe's Byerly Hall, and robotic snow removers for Harvard’s Facilities Maintenance Operations.
“This team of 21 undergraduate students did a spectacular job building from scratch an instrument that can address a real-world scientific problem,” said James G. Anderson, the Philip S. Weld Professor of Atmospheric Chemistry, who led the project. “They combined aspects of optical mechanical design, analog and digital electronics, and software systems design to come up with an instrument that is a major step forward in stratospheric observation technology.”
Anderson has been researching the link between chemistry and climate in the upper atmosphere for more than 40 years. It was his work that finally proved the connection between chlorofluorocarbons and halons that contain chlorine and bromine that catalytically destroys ozone over the polar regions in winter; research that has now turned to the question of ozone loss over the US in summer and its link to increased forcing of the climate by CO2 and CH4 from fossil fuel combustion worldwide.
On the first day of class, it was Anderson who gave the students — mostly electrical engineering and mechanical engineering concentrators — a crash course in atmospheric chemistry.
“That first day was really overwhelming,” said Simon Shuham, S.B. ’17, a mechanical engineering concentrator. “In order to tackle this problem, we had to not only understand the chemistry and the phenomena that causes ozone loss but how to research it on our own and communicate the science to others.”
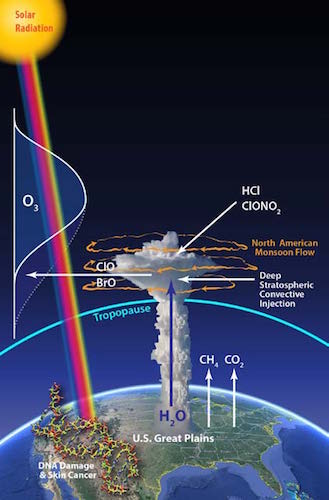 Stratospheric ozone — known as good ozone — absorbs most of the harmful ultraviolet sunlight traveling through the atmosphere. Research has shown that a one-percent decrease in stratospheric ozone can lead to a three-percent increase in skin cancer in humans. But this atmospheric barrier is at risk due to climate change, especially over the United States.
Stratospheric ozone — known as good ozone — absorbs most of the harmful ultraviolet sunlight traveling through the atmosphere. Research has shown that a one-percent decrease in stratospheric ozone can lead to a three-percent increase in skin cancer in humans. But this atmospheric barrier is at risk due to climate change, especially over the United States.
As violent summer storms increase in the U.S., more water is being pulled from the lower troposphere deep into the stratosphere. The additional water alters the properties of stratospheric particles, which react with HCl present in stratosphere to initiate a series of chemical reactions that destroy ozone. But little is known about the dynamics of this process because there hasn’t been an instrument to study it.
That was the challenge posed to the SEAS students. The instrument needed to be lightweight enough to be carried by a research balloon yet sturdy enough to withstand winds and rain from convective storms; operate at between 40,000 and 115,000 feet at temperatures ranging from minus 120 degrees to 120 degrees Fahrenheit; withstand the low pressure of the upper atmosphere; power itself and operate autonomously for a four- to six-week mission, all while measuring trace amounts of gases.
The prototype developed by the students meets all those requirements. Over the next year, the students who choose to remain on the project will work to transform the flight prototype into a NASA qualified flight instrument for launch into the stratosphere.
"The iteration process of making something that will fly is going to be really exciting,” said team member Carolina Pena, S.B. ’17, a mechanical engineering concentrator. “There is a lot more detail than you would expect, so we have to be able to juggle all those parts as a team.”
Working as a team was one of the biggest lessons of the course, said Daniel Wang, S.B. ’17, a mechanical engineering concentrator.
"None of us had worked on a team this big before,” he said. “But this is the norm for a lot of companies so this project really gave us a lot of real-world project experience.”
“This award will allow the student team to take their design from a classroom prototype to a full-fledged instrument that will help us understand how the chemistry in the stratosphere is changing in response to climate,” said Karena McKinney, a scientist at SEAS and one of the course Instructors. “The students have already made major advances in the instrument design and their results could make a major contribution to atmospheric science as well.”
Topics: Academics
Cutting-edge science delivered direct to your inbox.
Join the Harvard SEAS mailing list.
Scientist Profiles
James G. Anderson
Philip S. Weld Professor of Atmospheric Chemistry
Press Contact
Leah Burrows | 617-496-1351 | lburrows@seas.harvard.edu




